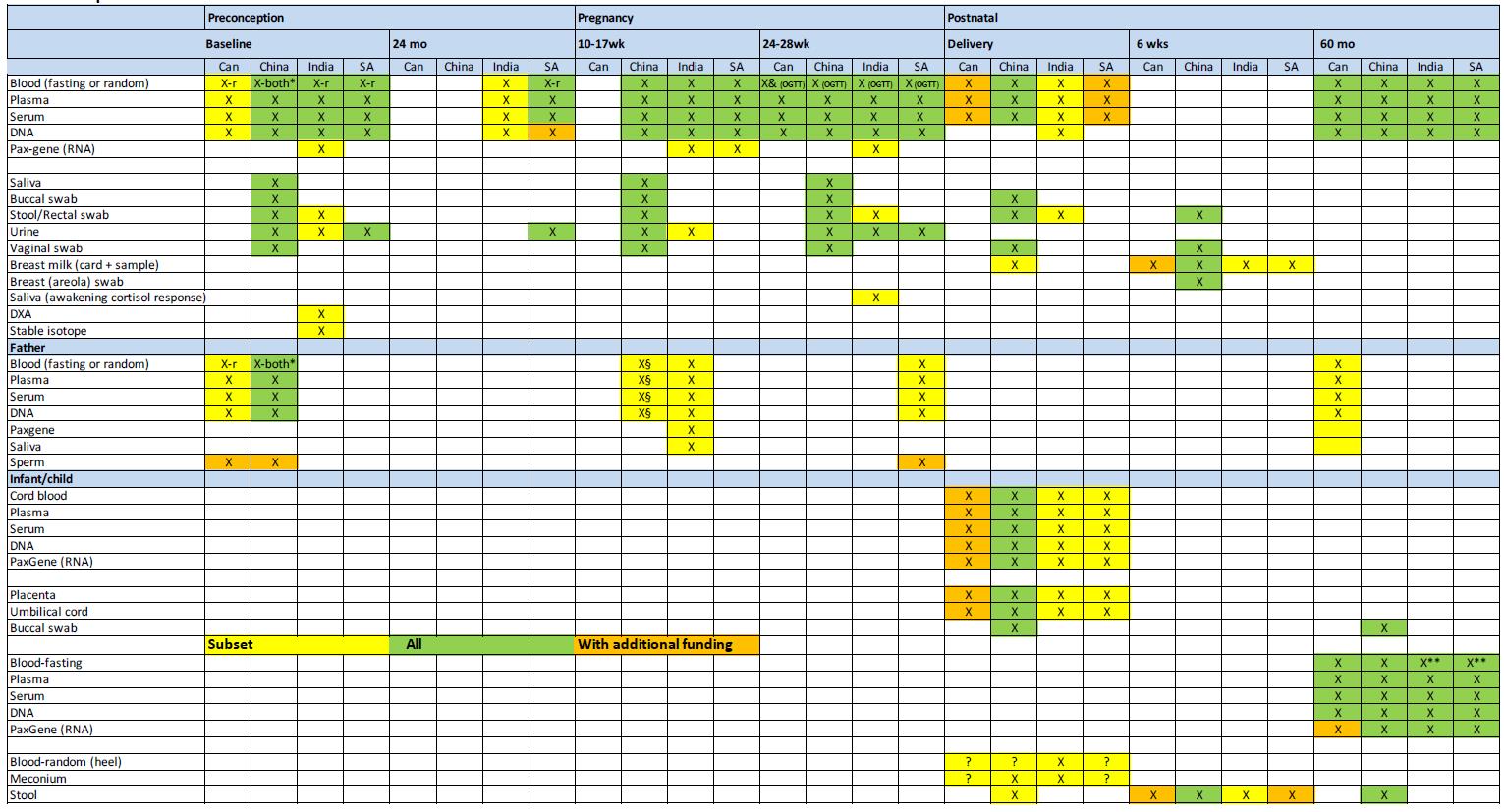
Biospecimens
The HeLTI Research Committee in collaboration with the HeLTI Biospecimen Working Group have agreed upon a harmonized selection of specific core biospecimens to be collected in all sites, including the principles governing the biospecimen collection, biobanking and access. While the HeLTI teams recognize the benefits of collecting multiple, longitudinal biospecimens, this must be balanced with country-specific cultural sensitivities, feasibility and cost. Emphasis has been placed on sample integrity over sample quantity. Numerous biospecimens will be collected in all study participants across sites; whereas other biospecimens will be taken only in a subset of participants. Additional biospecimen sampling will require approval from the HeLTI Research Committee as well as supplementary funding.
All biospecimens are collected, processed and aliquoted by trained technicians using established standard operating procedures (SOPs) used in other international cohort studies. Management of such samples takes place in a secure manner according to shared procedures and quality standards adopted by all teams to safeguard samples’ integrity. The assessment of measures and quality control of sample collection and storage is supported by an external international expert.
Overview of the Harmonised Biospecimen Data Collection Across the HeLTI Sites